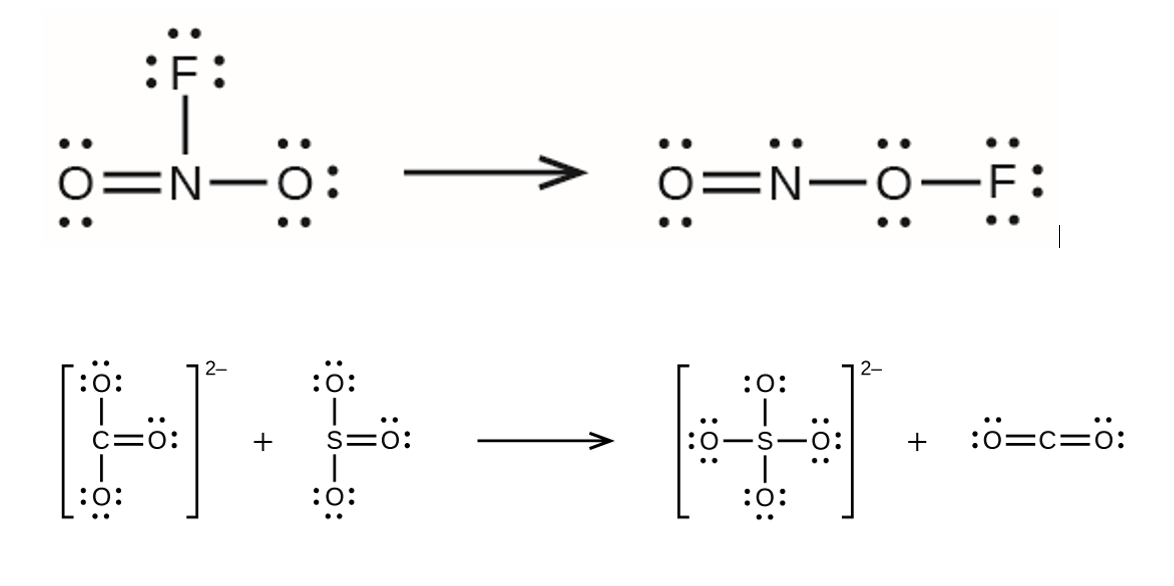
Review for Reaction Enthalpy Summative
Conservation of Mass Lab
The Law of Conservation of Mass states that mass can neither be created or nor destroyed but only converted during a chemical reaction. Thus, the total amount of matter does not change in a reaction. The reaction may have produced new material, but every atom of the original material is still present, just bonded into different material.
In your own words, what does the law of conservation of mass mean?
Answer here.
PURPOSE:
The purpose of this lab is to show visual evidence of the law of conservation of mass during a chemical reaction.
INTRODUCTION:
When chemical reactions occur, people tend to believe that matter is being destroyed when in fact matter is simply changing states. The Law of Conservation of Mass states that mass is neither created nor destroyed by chemical reactions or physical transformations. The mass of the products must equal the mass of the reactants. According to the law of conservation of mass, if a chemical reaction occurs in a closed system, there should be no difference in mass. In this lab the students will perform a chemical reaction and record data to see how the law of conservation of mass works for different systems.
Reaction I: Video https://www.youtube.com/watch?v=MUkb0qZeZCE
Mass BEFORE: ?
Mass AFTER: ?
Observations: Answer here.
REACTION II: Video https://www.youtube.com/watch?v=ruGa1qg6ltE
Mass Before Vinegar: ?
Mass Before Baking Soda: ?
Total Mass Before (Add the two reactants) ?
Mass After Reaction: ?
Observations Answer here.
ANALYSIS QUESTIONS:
1. What evidence to verify that there was a chemical reaction that occurred in both experiments?
Answer here.
2. How did the before mass of the systems compare with the after mass of the system for each trial?
Reaction I: Answer here. Reaction II: Answer here.
3. Did the reaction in Reaction I confirm or violate the law of conservation of mass? Explain using the data and observations.
Answer here.
4. Did the reaction in Reaction II confirm or violate the law of conservation of mass? Explain using the data and observation.
Answer here.
5. Both reactions produced carbon dioxide gas. Looking at the above reactions and understanding that the law of conservation of mass is a LAW that ALWAYS holds true. Why did it appear to be violated during part of this lab?
Answer here.
6. Explain how you could alter the previous procedure to ensure the law is demonstrated. Be sure to include why the new procedure would have an alternate affect on your mass. Be specific!
Answer here.
Conservation of Mass Lab
The Law of Conservation of Mass states that mass can neither be created or nor destroyed but only converted during a chemical reaction. Thus, the total amount of matter does not change in a reaction. The reaction may have produced new material, but every atom of the original material is still present, just bonded into different material.
In your own words, what does the law of conservation of mass mean?
Answer here.
PURPOSE:
The purpose of this lab is to show visual evidence of the law of conservation of mass during a chemical reaction.
INTRODUCTION:
When chemical reactions occur, people tend to believe that matter is being destroyed when in fact matter is simply changing states. The Law of Conservation of Mass states that mass is neither created nor destroyed by chemical reactions or physical transformations. The mass of the products must equal the mass of the reactants. According to the law of conservation of mass, if a chemical reaction occurs in a closed system, there should be no difference in mass. In this lab the students will perform a chemical reaction and record data to see how the law of conservation of mass works for different systems.
Reaction I: Video https://www.youtube.com/watch?v=MUkb0qZeZCE
Mass BEFORE: ?
Mass AFTER: ?
Observations: Answer here.
REACTION II: Video https://www.youtube.com/watch?v=ruGa1qg6ltE
Mass Before Vinegar: ?
Mass Before Baking Soda: ?
Total Mass Before (Add the two reactants) ?
Mass After Reaction: ?
Observations Answer here.
ANALYSIS QUESTIONS:
1. What evidence to verify that there was a chemical reaction that occurred in both experiments?
Answer here.
2. How did the before mass of the systems compare with the after mass of the system for each trial?
Reaction I: Answer here. Reaction II: Answer here.
3. Did the reaction in Reaction I confirm or violate the law of conservation of mass? Explain using the data and observations.
Answer here.
4. Did the reaction in Reaction II confirm or violate the law of conservation of mass? Explain using the data and observation.
Answer here.
5. Both reactions produced carbon dioxide gas. Looking at the above reactions and understanding that the law of conservation of mass is a LAW that ALWAYS holds true. Why did it appear to be violated during part of this lab?
Answer here.
6. Explain how you could alter the previous procedure to ensure the law is demonstrated. Be sure to include why the new procedure would have an alternate affect on your mass. Be specific!
Answer here.
